Medical Devices – Quality Control & Cybersecurity requirements and the role of FDA
E-health has advanced for many years. Over the following few years, networkization will increase. e-Health organizations are connecting medical equipment more and more. Cybersecurity will consequently become more significant. With its 510(k) process (premarket control), the FDA (Food Drug Administration) has established quality standards with the intention of having a tool to standardize the quality of the devices entering the market while also having the option to remove devices from the market should the devices harm patients (post market control). Additionally, FDA has provided guidelines for how Cybersecurity should be taken into account during the design of medical devices. But it appears that this is not enough.
To address this issue, we have created a unique process that incorporates various best practices to produce high-quality, secure products for the market:
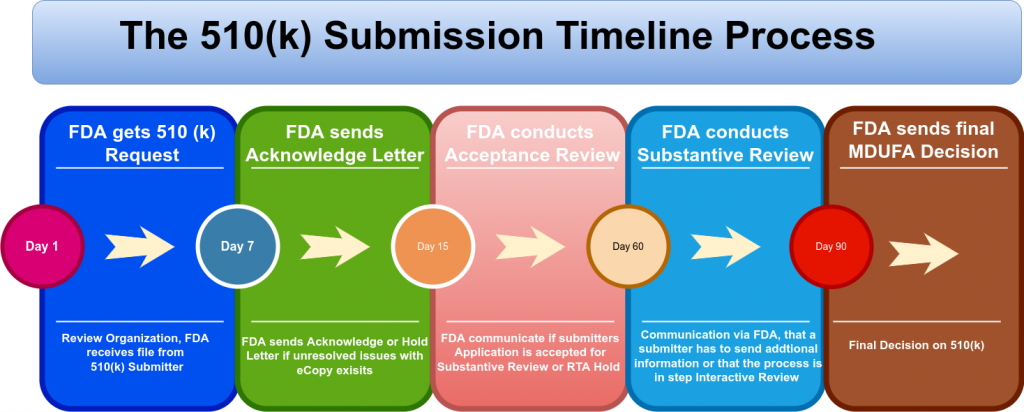
01.
Follow 510 (k)
Follow process, goals & quality aspects.
02.
Cybersecurity Requirements
Follow Best Practices & Principle Security by Design.
03.
Assessment / Gaps
Review and develop the Gaps further.
05.
Reassessment within Postmarked Phase
Implement suggested mitigation steps and follow implementations steps.
04.
Implement within Premarket Phase
Implement suggested mitigation steps and follow Reassessment.
06.
Maintenance & Continuous Testing & Lessons Learned
Follow appropriate steps in order to enrich product quality, test to identify defects as early as possible and to improve overall organisation and understanding for future projects.
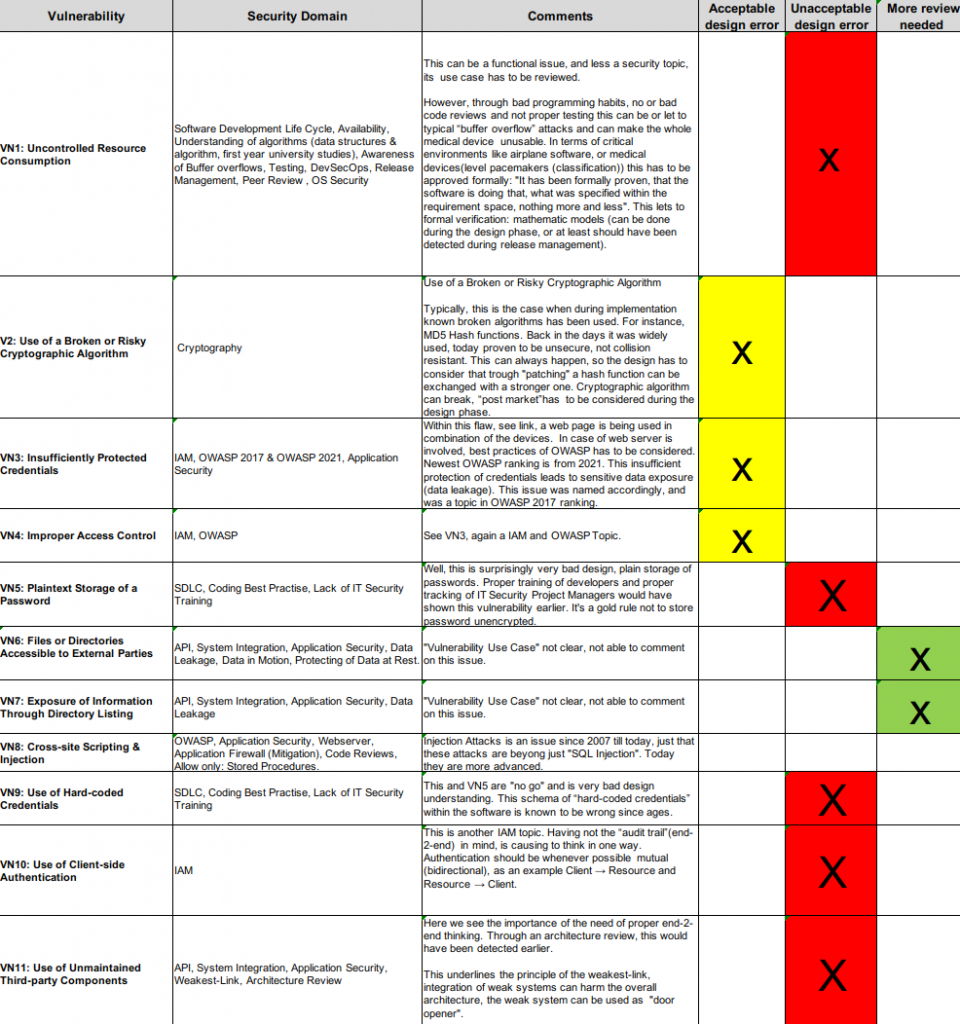
Several medical gadgets have been examined by Blueguard for faults. As a consequence of this analysis, it has been determined that even the intricate 510(k) process and the Cybersecurity guideline are insufficient to ensure the security of medical device construction. To demonstrate this point, we choose the manufacturer Fresenius Kabi as our example, and we categorize their design flaws into acceptable and unacceptable categories.
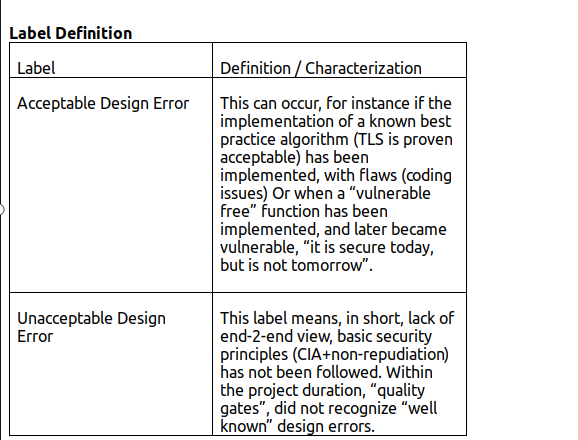
After reviewing each vulnerability, we come to the conclusion as to why security was not considered during the premarket process and which security area has been overlooked.
Review on Vulnerabilities & Findings
Blueguard can help with the 510(k) process to obtain the FDA’s necessary approval. We will help with the design of your medical device product and go above the FDAs Cybersecurity requirements.